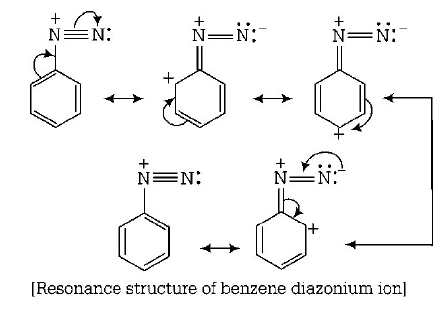
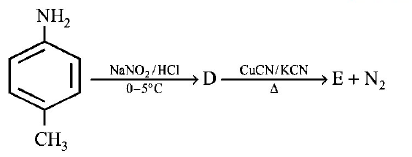
41. Which of the following will be most stable diazonium salt $$RN_2^ + {X^ - }?$$
A
$$C{H_3}N_2^ + {X^ - }$$
B
$${C_6}{H_5}N_2^ + {X^ - }$$
C
$$C{H_3}C{H_2}N_2^ + {X^ - }$$
D
$${C_6}{H_5}C{H_2}N_2^ + {X^ - }$$
Answer :
$${C_6}{H_5}N_2^ + {X^ - }$$
42. Aniline when diazotised in cold and then treated with $$N, N$$ -dimethylaniline gives a coloured product. The structure of this product is
A
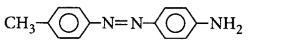

B


C


D
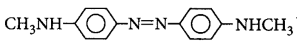
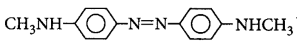
Answer :


43. Butanonitrile may be prepared by heating :
A
Propyl alcohol with $$KCN$$
B
Butyl alcohol with $$KCN$$
C
Butyl chloride with $$KCN$$
D
Propyl chloride with $$KCN$$
Answer :
Propyl chloride with $$KCN$$
44. Which of the following is more basic than aniline?
A
Diphenylamine
B
Triphenylamine
C
$$p$$ - nitroaniline
D
Benzylamine
Answer :
Benzylamine
45. Oxidation of aniline with manganese dioxide and sulphuric acid produces
A
phenyl hydroxylamine
B
nitrobenzene
C
$$p$$-benzoquinone
D
phenol
Answer :
$$p$$-benzoquinone
47. The most reactive amine towards dilute hydrochloric acid is _________.
A


B
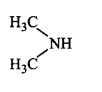

C
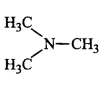

D
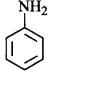

Answer :


48. Amine that cannot be prepared by Gabrielphthalimide synthesis is
A
aniline
B
benzyl amine
C
methyl amine
D
$$iso$$ - butylamine
Answer :
aniline
49. A compound $$(X)$$ with molecular formula $${C_3}{H_9}N$$ reacts with $${C_6}{H_5}S{O_2}Cl$$ to give a solid which is insoluble in alkali. $$(X)$$ is
A
$$C{H_3}C{H_2}C{H_2}N{H_2}$$
B
\[\underset{\begin{smallmatrix}
\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,| \\
\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,C{{H}_{3}}
\end{smallmatrix}}{\overset{\begin{smallmatrix}
\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,C{{H}_{3}} \\
\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,|
\end{smallmatrix}}{\mathop{C{{H}_{3}}-N}}}\,\]
C
$$C{H_3} - NH - C{H_2}C{H_3}$$
D
\[C{{H}_{3}}\underset{\begin{smallmatrix}
|\,\,\,\,\, \\
C{{H}_{3}}\,\,
\end{smallmatrix}}{\mathop{-CH-}}\,N{{H}_{2}}\]
Answer :
$$C{H_3} - NH - C{H_2}C{H_3}$$
50. Choose the correct statement from the following.
A
Methyl amine is less soluble than propyl amine.
B
Methyl amine is solid with fishy odour.
C
Aniline gets coloured on storage.
D
Amines are more polar than alcohols.
Answer :
Aniline gets coloured on storage.