41.
On vigorous oxidation by permanganate solution.
$${\left( {C{H_3}} \right)_2}C = CH - C{H_2} - CHO$$ gives
A
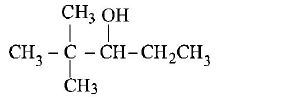
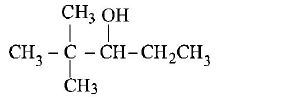
B
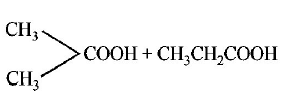
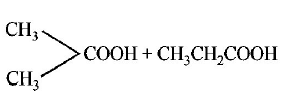
C
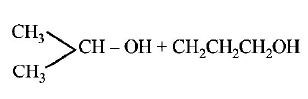
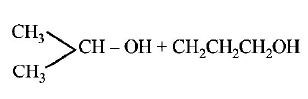
D
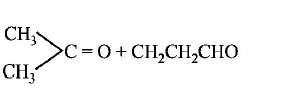
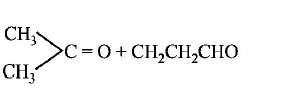
Answer :
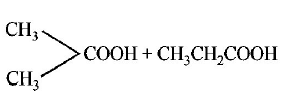
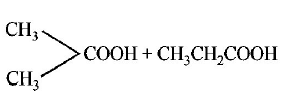
42.
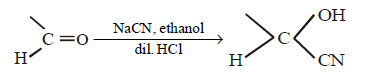
What is the product of the following reaction ?
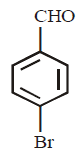 \[\xrightarrow[dil.\,HCl]{NaCN,\text{ethanol}}\]
\[\xrightarrow[dil.\,HCl]{NaCN,\text{ethanol}}\]
A
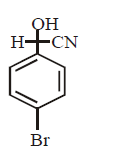

B
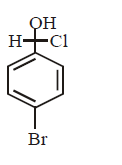

C
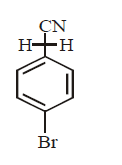

D
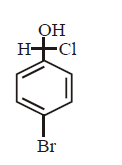

Answer :


43. Trichloroacetaldehyde was subjected to Cannizzaro’s reaction by using $$NaOH.$$ The mixture of the products contains sodium trichloroacetate and another compound. The other compound is :
A
2, 2, 2 - Trichloroethanol
B
Trichloromethanol
C
2, 2, 2 - Trichloropropanol
D
Chloroform
Answer :
2, 2, 2 - Trichloroethanol
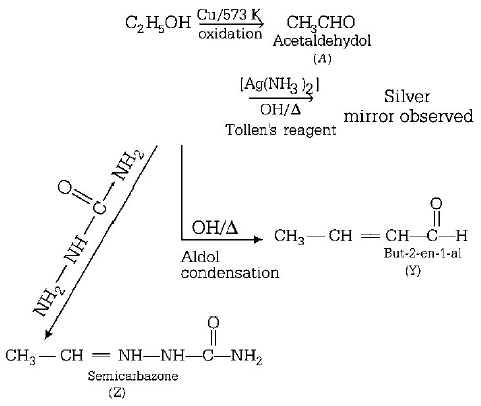
44.
Consider the reactions,

Identify $$A, X,Y$$ and $$Z$$
A
$$A$$ - methoxymethane, $$X$$ - ethanoic acid, $$Y$$ - acetate ion, $$Z$$ - hydrazine
B
$$A$$ - methoxymethane, $$X$$ - ethanol, $$Y$$ - ethanoic acid, $$Z$$ - semicarbazide
C
$$A$$ - ethanal, $$X$$ - Acetaldelyde, $$Y$$ - but - 2 - enal, $$Z$$ - semicarbazone
D
$$A$$ - ethanol, $$X$$ - acetaldehyde, $$Y$$ - butanone, $$Z$$ - hydrazone
Answer :
$$A$$ - ethanal, $$X$$ - Acetaldelyde, $$Y$$ - but - 2 - enal, $$Z$$ - semicarbazone
45. Hydrocarbons are formed when aldehydes and ketones are reacted with amalgamated zinc and conc. $$HCl.$$ The reaction is called
A
Cannizzaro reaction
B
Clemmensen reduction
C
Rosenmund reduction
D
Wolff-Kishner reduction
Answer :
Clemmensen reduction
46.
Predict the products in the given reaction,
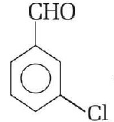 \[\xrightarrow{{50\% \,\,KOH}}\]
\[\xrightarrow{{50\% \,\,KOH}}\]
A


B
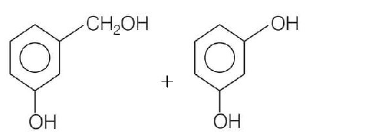

C
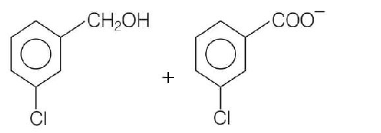

D
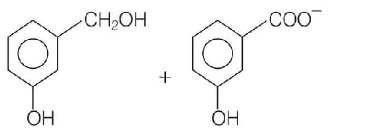

Answer :


47.
Fill in the blanks by suitable choices.
The carbon atom in carbonyl group is ______ hybridised. The carbonyl group in aldehydes and ketones undergoes ______ reactions. Aldehydes which have $$\alpha {\text{ - }}H$$ atom undergo ______ reaction while aldehydes which have no $$\alpha {\text{ - }}H$$ atom undergo ______ reaction.
A
$$s{p^3},$$ nucleophilic substitutions, aldol condensation, Cannizzaro
B
$$sp,$$ electrophilic substitution, Cannizzaro, aldol condensation
C
$$s{p^2},$$ nucleophilic addition, aldol condensation, Cannizzaro
D
$$s{p^3},$$ electrophilic addition, Cannizzaro, aldol condensation
Answer :
$$s{p^2},$$ nucleophilic addition, aldol condensation, Cannizzaro
48. Which of the following is incorrect ?
A
\[FeC{{l}_{3}}\] is used in detection of phenols.
B
Fehling solution is used in detection of glucose.
C
Tollen’s reagent is used in detection of unsaturation.
D
\[NaHS{{O}_{3}}\] is used in detection of carbonyl compounds.
Answer :
Tollen’s reagent is used in detection of unsaturation.
49. A substance $${C_4}{H_{10}}O$$ yields on oxidation a compound, $${C_4}{H_8}O$$ which gives an oxime and a positive iodoform test. The original substance on treatment with $$conc.\,{H_2}S{O_4}$$ gives $${C_4}{H_8}.$$ The structure of the compound is
A
$$C{H_3}C{H_2}C{H_2}C{H_2}OH$$
B
$$C{H_3}CHOHC{H_2}C{H_3}$$
C
$${\left( {C{H_3}} \right)_3}COH$$
D
$$C{H_3}C{H_2} - O - C{H_2}C{H_3}$$
Answer :
$$C{H_3}CHOHC{H_2}C{H_3}$$
50. Few simple chemical tests are given below to differentiate between the pairs of compounds. Which of the following tests is not correct for differentiation?
A
Propanal and propanone - Silver mirror test
B
Acetophenone and benzophenone - Iodoform test
C
Ethanal and propanal - Fehling's test
D
Benzoic acid and ethyl benzoate - Sodium bicarbonate test
Answer :
Ethanal and propanal - Fehling's test