221. The correct order of decreasing acidic strength of trichloroacetic acid $$(A),$$ trifluoroacetic acid $$(B),$$ acetic acid $$(C)$$ and formic acid $$(D)$$ is
A
$$B > A > D > C$$
B
$$B > D > C > A$$
C
$$A > B > C > D$$
D
$$A > C > B > D$$
Answer :
$$B > A > D > C$$
222. The correct representation of 4-hydroxy-2-methylpent-2-en-1-al is
A
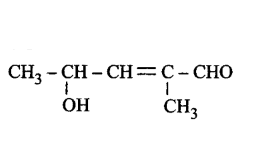

B


C
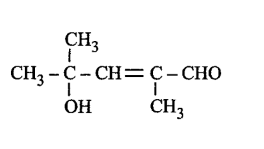
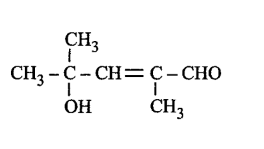
D
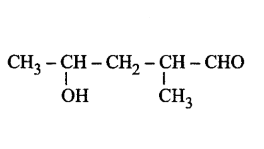
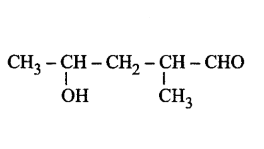
Answer :


223. Allyl isocyanide has :
A
$$9\sigma \,{\text{and}}\,4\pi \,{\text{bonds}}$$
B
$$8\sigma \,{\text{and}}\,5\pi \,{\text{bonds}}$$
C
$$9\sigma ,3\pi \,{\text{and}}\,2\,{\text{non - }}\,{\text{bonds}}\,{\text{electrons}}$$
D
$$8\sigma ,3\pi \,{\text{and}}\,4\,{\text{non - }}\,{\text{bonds}}\,{\text{electrons}}$$
Answer :
$$9\sigma ,3\pi \,{\text{and}}\,2\,{\text{non - }}\,{\text{bonds}}\,{\text{electrons}}$$
224. The IUPAC name for \[C{{H}_{3}}-\overset{\begin{smallmatrix} O \\ \parallel \end{smallmatrix}}{\mathop{C}}\,-C{{H}_{2}}-C{{H}_{2}}-\overset{\begin{smallmatrix} O \\ \parallel \end{smallmatrix}}{\mathop{C}}\,-OH\] is _________.
A
1-hydroxypentane-1, 4-dione
B
1, 4-dioxopentanol
C
1-carboxybutan-3-one
D
4-oxopentanoic acid
Answer :
4-oxopentanoic acid
225. Geometrical isomers differ in
A
position of functional group
B
position of atoms
C
spatial arrangement of atoms
D
length of carbon chain
Answer :
spatial arrangement of atoms
226. How many chain isomers could be obtained from the alkane, $${C_6}{H_{14}}?$$
A
Four
B
Five
C
Six
D
Seven
Answer :
Five
227.
The IUPAC name of the compound having formula 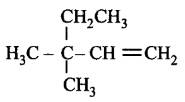 is
is
A
3, 3, 3-trimethylprop-1-ene
B
1, 1, 1-trimethylprop-2-ene
C
3, 3-dimethylpent-1-ene
D
2, 2-dimethylbut-3-ene
Answer :
3, 3-dimethylpent-1-ene
228. The distillation technique most suited for separating glycerol from spent-lye in the soap industry is :
A
Steam distillation.
B
Distillation under reduced pressure.
C
Simple distillation
D
Fractional distillation
Answer :
Distillation under reduced pressure.
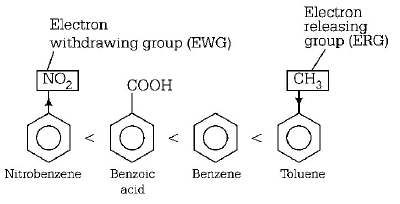
229. Among the following compound one that is most reactive towards electrophilic nitration is
A
benzoic acid
B
nitrobenzene
C
toluene
D
benzene
Answer :
toluene
230. Which of the following would react most readily with nucleophiles ?
A
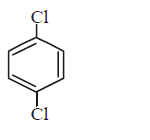

B
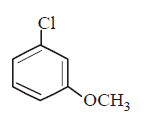

C


D
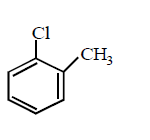

Answer :