Question
Choose the correct statement about the given figures.
Choose the correct statement about the given figures.
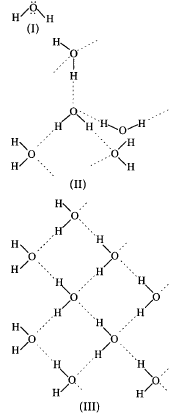
A.
(II) represents solid state while (III) represents liquid state.
B.
(II) represents liquid state while (III) represents solid state.
C.
(I) represents solid state while (III) represents liquid state.
D.
(I) represents liquid state while (III) represents solid state.
Answer :
(II) represents liquid state while (III) represents solid state.
Solution :
In liquid state water molecules are hydrogen bonded while in solid state water molecules are arranged in tetrahedral manner with open cage structure.
In liquid state water molecules are hydrogen bonded while in solid state water molecules are arranged in tetrahedral manner with open cage structure.